Published by Zdeněk HRADÍLEK, Petr MOLDŘÍK, Roman CHVÁLEK
VŠB – Technical University of Ostrava, Department of Electrical Power Engineering
Abstract. Attention is paid to the electrical energy storage systems that are already used in the framework of electrical power system, and further to the systems that are studied and developed for this purpose. The described storage systems should work especially in synergy with unreliable renewable energy sources, such as wind and photovoltaic power plants. Those use the renewable source that gains ground quickly not only in the Czech Republic. Both the systems that make it possible to ensure high charging and discharging rates for a short time and the systems of great storage capacity that are able to store and transmit electrical energy for more hours are described.
Streszczenie. Skupiono się na systemach magazynowania energii elektrycznej obecnie stosowanych w ramach funkcjonowania systemu elektroenergetycznego, a w następnej kolejności na systemach będących w fazie badań i projektów. Opisane systemy magazynowania powinny współdziałać w szczególności z niestabilnymi źródłami odnawialnymi, jak wiatr i elektrownie fotowoltaiczne. Taki sposób użytkowania źródeł odnawialnych szybko ugruntowuje się nie tylko w Republice Czeskiej. Opisano zarówno systemy zapewniające wysoki wskaźnik ładowania i rozładowania w krótkim czasie, jak i systemy o wielkich zdolnościach magazynowych, które są w stanie przechowywać i przekazywać energię elektryczną przez wiele godzin. (Systemy magazynowania energii elektrycznej).
Keywords: energy storage, renewable source, storage system.
Słowa kluczowe: magazynowanie energii, źródło odnawialne, system magazynowania.
Introduction
There is a rising number of applications of renewable energy sources (RES) within electric power systems (EPS). Speaking of the Czech Republic, those are especially photovoltaic and wind power plants that are characteristic for their electric energy supply capacity variable and unreliable over time, fully dependant on weather conditions. These characteristics imply the need to establish a certain power backups to cater for blackout conditions and this backup concerns their full installed capacity. Problems associated with operation of these plants can be solved by means of storage of the electrical energy they produce. There are numerous different storage technologies.
Energy Storage Systems
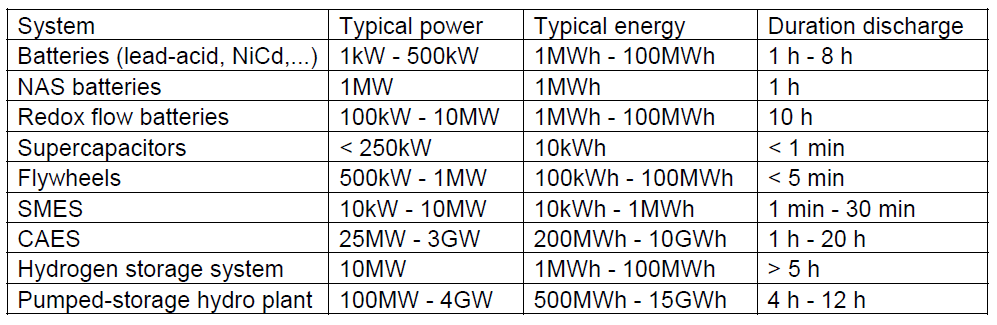
Systems for storage of electrical energy can be divided into two basic categories. The first one includes those systems to enable high charging and discharging performance for a short period of time. There are Supercapacitors, Superconductive magnetic accumulators (SMES) and rotary Flywheel accumulators. The second category then includes systems with great storage capacity able to store and transmit electrical energy for several hours. This category comprises Batteries, Compressed air accumulators (CAES), Redox flow batteries, Pumped storage hydro plants, and the last but not the least are the Hydrogen storage systems – the system of electrolyzer and a fuel cells.
Battery Energy Storage (Batteries)
These accumulators transform the chemical energy to electric power and vice versa. They comprise secondary cells, which are similar to the primary units (single discharge only) with the limited amount of reactants. However, the reaction products developed during cell discharge can be transformed back into initial active reactants, with the external electric power feed, which will re-instate the cell´s charged condition. The battery´s ability to supply power is then limited by its internal resistance, which increases with the cell ageing. There is a similar limit on the charging current, which implies the long period needed for charging.
The most common batteries currently are the lead-acid, NiCd, NiMH and Li-ion cells, different with the electrolyte and electrode materials used. The life span of most batteries ranges within hundreds of charge/discharge cycles. Apart from the charging method, the life span is also strongly affected by the operational temperature. Batteries will not be very suitable for environments requiring strong impulse current, these would rather work under constant loads. These are mainly used in vehicles, consumer electronics and UPS units. [1]
Sodium-Sulfur (NAS) Batteries
NAS batteries are high capacity battery systems developed for electric power applications. This battery consists of liquid (molten) sulfur at the positive electrode and liquid (molten) sodium at the negative electrode as active materials separated by a solid beta alumina ceramic electrolyte. The electrolyte allows only the positive sodium ions to go through it and combine with the sulfur to form sodium polysulfides (2Na + 4S = Na2S4). [2]
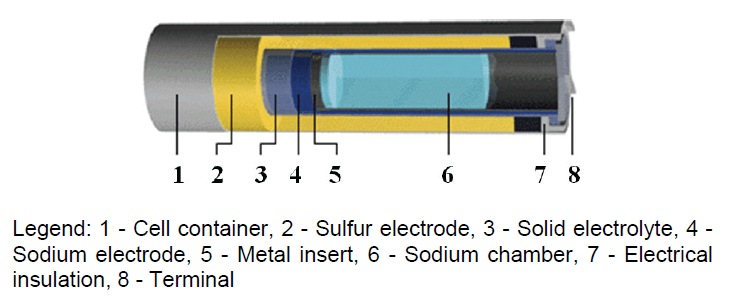
During discharge, as positive Na+ ions flow through the electrolyte and electrons flow in the external circuit of the battery producing about 2 volts. This process is reversible as charging causes sodium polysulfides to release the positive sodium ions back through the electrolyte to recombine as elemental sodium. This hermetically sealed battery is kept at approximately 300°C and is operated under conditions such that the active materials at both electrodes are liquid and the electrolyte is solid. At this temperature, since both active materials react rapidly and because the internal resistance is low, the NAS battery performs well. Because of reversible charging and discharging the NAS battery can be used continuously. Efficiency of NAS battery cells is about 89%. [2]
Redox Flow Batteries
The reduction-oxidation (Redox) flow batteries are considered highly prospective, as these are able to store large amounts of electric power. For this purpose, the ability of some chemical elements (e.g. vanadium) to have more valence states is utilized. The core of this system comprises a reversible reduction-oxidation cell to accommodate the transformation of electrical energy into chemical energy, bound within the electrolyte. These Redox batteries allow for continuous exchange of electrolyte. This ensures their continuous operation until the electrolyte supply has been used up. Electrolytes circulate within two circuits separated by the ion exchanger membrane inside the very cell. The cell interior provides for oxidizing of one form of the electrolyte upon electrochemical process, with the other one being subject to reduction due to the electric current fed or drained into the external electric circuit with the use of electrodes (see Figure 2).
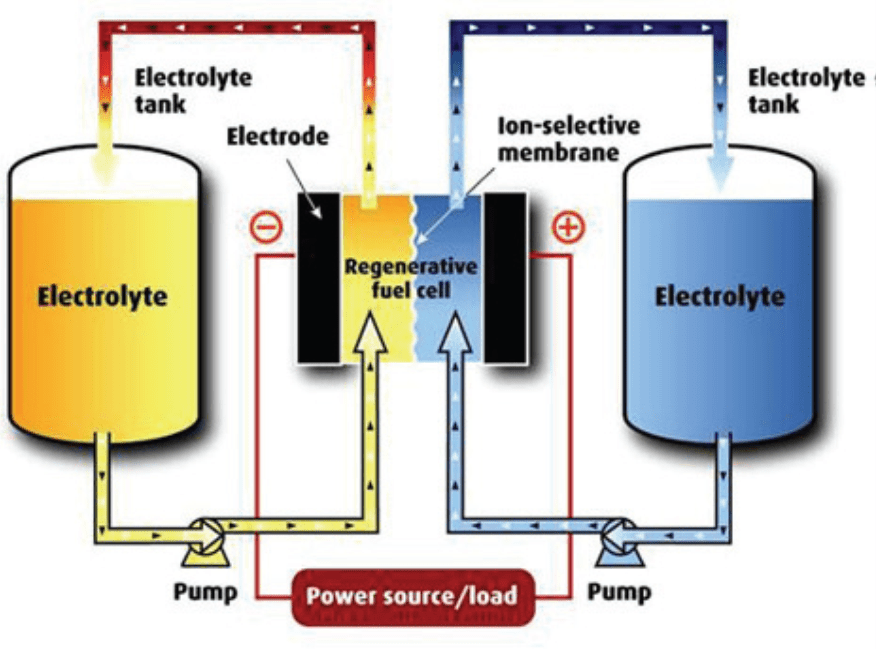
The storage capacity is determined by the amount of electrolyte in store tanks, while the actually achievable volume energetic density of electrolyte for a full charging (discharging) cycle is listed within the range of 15 to 25 kWh/m3. The unit does not show any performance or capacity reduction after more than 12 thousand charging cycles, the estimated life span of membranes is approximately 15 years. [3]
Supercapacitors
Also referred to as super condensers, are actually electrolytic capacitors with high capacity of thousands of F (Farad) and the ability for rapid charging and discharging. The electrodes are made from special materials as the micro-porous active carbon, which features extreme surface properties of up to 2000 m2/g and the gap of several nanometres between particular charged layers. These electrodes are separated with the polypropylene foil, the space within is filled with liquid electrolyte. The operation voltage is approx. 2,5V. Any higher voltage can be achieved by series setup of basic cells. The low internal resistance value enables for rapid discharge, the excellent performance provided by super capacitor achieves the values of several kW per 1kg of weight. Their electric parameters are maintained even under low temperatures down to -40°C. Supercapacitors represent ideal units to be used in applications with the need for peak current supply for a limited period of time. Their commercial utilisation has been launched recently only. [3]
High-Speed Flywheels
There are still the more common low-speed flywheels (rotating by up to approx. 7.000 rpm) provided with steel rotors. The very strong composite materials allow for development of light high-speed flywheels with the maximum speed up to approx. 50.000 rpm. To reduce the friction produced, the rotor will be housed within vacuum with magnetic uplift. The rotor also comprises permanent magnets, which help with its initial roll-up or generate current within coils.
These units also feature sophisticated electronics to ensure the safe and maintenance free operation. The contemporary flywheels can provide the performance from several kW up to approx. 1 MW. Their advantage lies in the optional operation of several units in parallel. A steel flywheel can provide approx. 200 kW of backup power within a 3-phase 400V network with the revolutions range of 7.700 – 4.000 rpm. Any such unit can be also operated separately as a short-term backup or a component within a larger system. [5]
Superconducting Magnetic Energy Storage (SMES)
The superconducting magnetic electrical energy storage units enable the rapid absorption and supply of power without any limitations or losses. Energy is stored within the magnetic field of coil bearing current, this coil being housed within a cryostat. Such coil is made from a superconductor in order to eliminate any resistance losses. However, there are power losses incurred during operation of the cooling unit, which maintains the coil superconductor below the critical temperature level.
The SMES system helps to store up to thousands of MWh and it can be used to balance peaks of electric power take-off. The very superconducting coil, depending on the size and method of use, can be made of a solenoid toroid. The superconductor energy accumulator might be the alternate solution for electrical energy storage in forthcoming years. However the need for cooling to preserve its operational temperature below critical level makes this unit costly compared to other technologies. [5]
CAES (Compressed Air Energy Storage) System
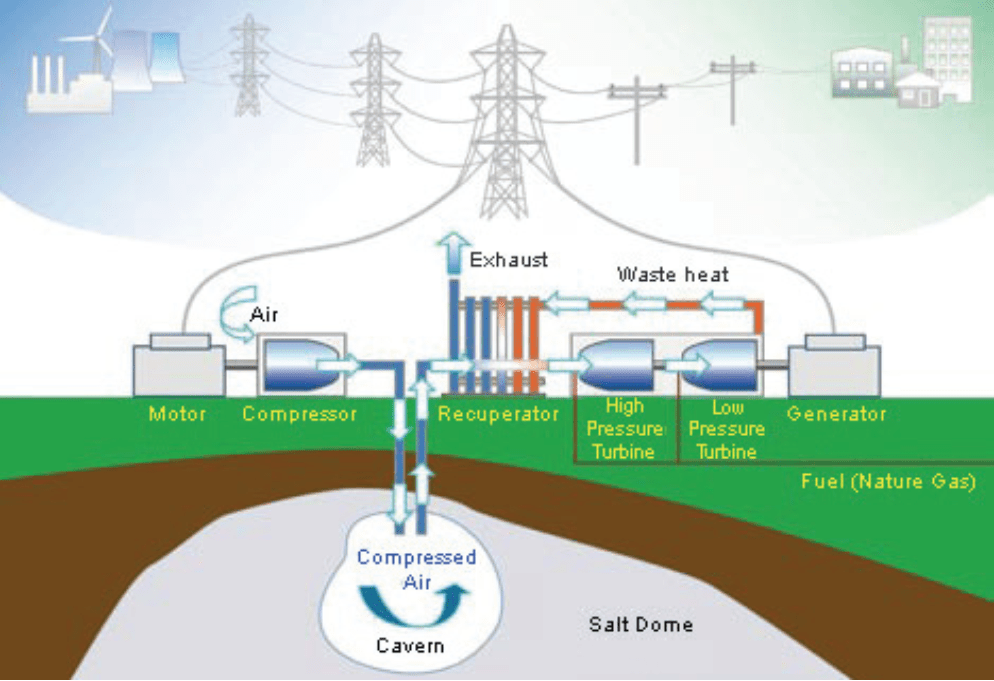
This is a compressed air accumulator linked with a turbine to supply power during take-off peaks within the network as needed. It is a peaking gas turbine power plant that consumes less than 40% of the gas used in conventional gas turbine to produce the same amount of electric output power. This is because, unlike conventional gas turbines that consume about 2/3 of their input fuel to compress air at the time of generation, CAES pre-compresses air using the low cost electricity from the electrical power system at off-peak times and utilizes that energy later along with some gas fuel to generate electricity as needed. The compressed air is often stored in appropriate underground mines or caverns created inside salt rock (see Figure 3). The CAES technology is still used very rarely in global commercial projects. [6]
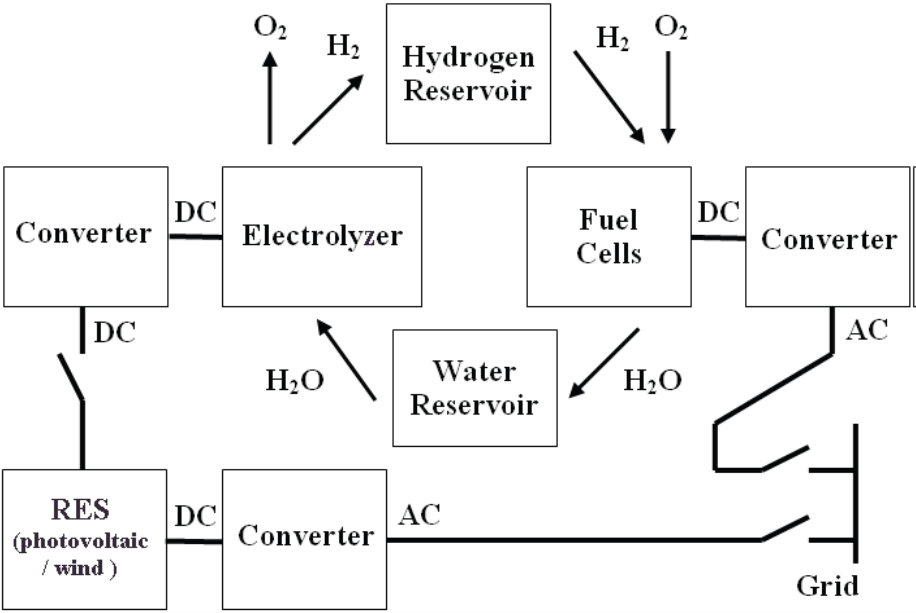
Hydrogen Storage System
The main parts of this system comprise the hydrogen generator, the so called “electrolyzer”, the magazine to hold the hydrogen produced, the demineralised water magazine and the hydrogen fuel cells. Further components necessary include the semiconductor invertors, compressors and vents. The cooperation between the hydrogen system and the non-controlled RES is based on the principle that the power produced by RES will be used to produce hydrogen within the period of lower EPS workload and this hydrogen will be stored. The hydrogen in storage will be later used in fuel cells, consumed during production of electric energy in the period associated with demand for electric energy by EPS. Figure 4 shows the diagram of hydrogen system.
Pumped-Storage Hydro Plant
A pumped-storage hydro plant (PSHP) represents a system able to store large amounts of electric energy. PSHP units are used to cater for peak demands for electric energy within EPS. Apart from the latter, these units also play their unique role within the control of output of the national energetic system in terms of emergency reserve. Their installed capacity within the territory of Czech Republic amounts to 1.175 MW. The PSHP serves for storage of electric energy using the potential gravity energy of water. The unit comprises of two reservoirs, whereas one of them is placed below the other. These reservoirs are linked with gravity piping of large diameter. During the period of power surplus within the EPS (at night), this power is used to pump the water from the bottom reservoir up into the top one. Once the ESP has developed a demand for large amount of peak power, this water will be subject to controlled discharge from the top reservoir down into the bottom one via the hydro plant turbine. This storage system is costly and requires significant landscape adaptations. The rate of efficiency of this pumping cycle is approx. 75%.
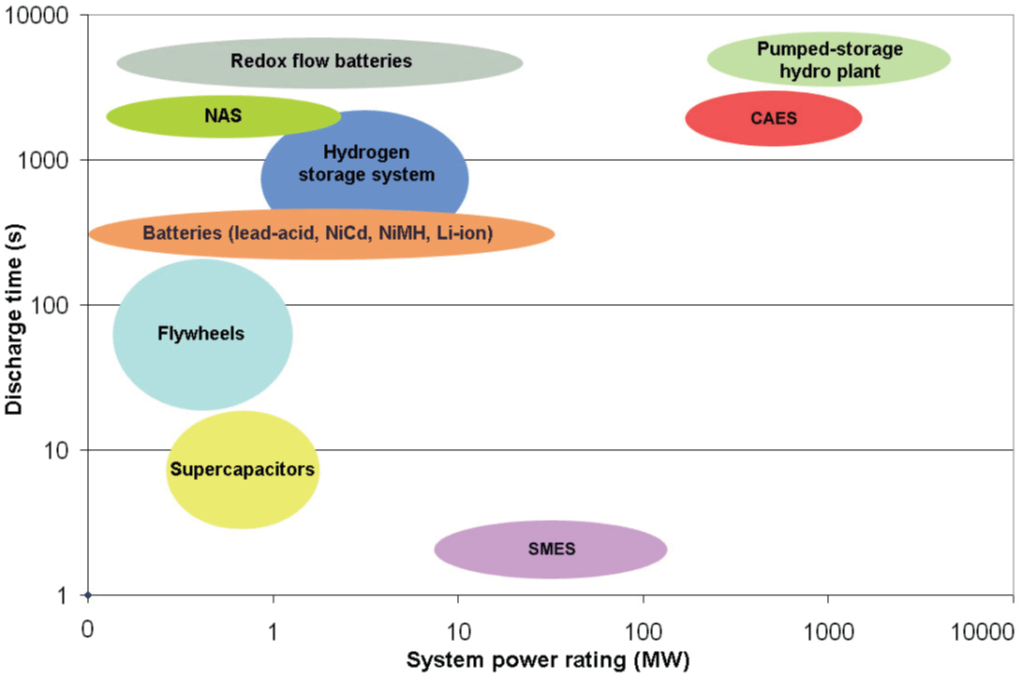
Storage Systems Benchmark
The storage systems described above have been grouped within the Table 1 to allow for proper comparison of basic parameters. Figure 5 then shows a graphic comparison of these systems, in order corresponding to their power rating and the discharge time.
Laboratory Research into Hydrogen Storage System
The storage system based on the hydrogen technology seems to be very prospective with regard to cooperation with the non-controlled RES. There is currently a research focused on this technology in progress, together with examination of its practical application. This research also involves our laboratory at the Department of Electrical Power Engineering, VŠB – TU Ostrava, concerned with sophisticated experimental laboratory implementation of the model hydrogen system for storage of electric energy produced by photovoltaic panels. Further detailed information about these panels is contained within other paper by authors dealing with the issue of photovoltaic power plant operation within the territory of the Czech Republic.
Our research concerns testing and measuring of particular components of the hydrogen system designed in order to optimise their operation to provide reliable, safe and highly efficient units. See the block diagram in Figure 4.
Table 1. Basic parameters of storage systems [7]
Electrolyzer
Electrolyzer producing hydrogen is the first one of the most important parts of the entire hydrogen system. The process of electrolysis helps use the energy to decompose water to the very elements: hydrogen and oxygen. The electrolyzer comprises a series of cells equipped with positive and negative electrodes respectively, these are dipped in water. The level of conductivity is achieved by addition of hydrogen or hydroxyl ions (hydroxides), the most common form is the alkali potassium hydroxide (KOH). The amount of hydrogen produced depends on the flow density. The current electrolyzers feature energetic efficiency between 65 and 80 %. We use the Hogen GC600 electrolyzer comprising of electrodes and the gas separator to provide for separation of hydrogen produced from oxygen.
Electrolyzers of this kind use the sulhponated tetrafluorethylene (Nafion) to substitute the liquid electrolyte. The input of this electrolyzer is 1.100W, its operation temperature ranges around 85°C, the volume of hydrogen produced equals to 0,6 l/min (high purity: 99,999%), the output pressure of hydrogen reaches up to 13,8 bar. The source of energy used for decom-position of water is represented by photovoltaic panels.
Despite the low specific density, hydrogen has the highest ratio of energy to weight among all the fuels. As far as gases are concerned, its density is the lowest possible and it also features the second lowest boiling point of all the known substances. These properties then determine the options for its storage. [8]
It can be stored as a highly compressed gas, as a liquid in cryogenic magazines or as a bonded gas (e.g. in metal-hydrides). The storage of hydrogen in gaseous form requires large magazine volumes and high compression. High pressure hydrogen storage units represent the most common method used. If liquid, the hydrogen can be stored below its boiling point only, which is equal to 20 K (-253 °C). Owing to the latter, the liquefying of hydrogen is a strongly energy demanding process. The storage systems with metal-hydrides are based on the principle of easy absorption of gas by certain materials under high pressure and moderate temperatures. These substances would then release hydrogen, when beating heated under low pressure and relatively high temperatures. [8]
We use metal-hydride bottles and pressure cylinders for laboratory research.
PEM Fuel Cell
A fuel cell is a device, which uses the electrochemical reaction to transform chemical energy held by the fuel (hydrogen), aided by the oxidizing agent, to electric power, water and heat. This transformation occurs within catalytic reactions on electrodes and it is mainly based on reversed principle of water electrolysis. This efficiency of this energy production process is up to 60 % (under laboratory conditions), the real value would then be between 35 and 50 % (depending on the load and fuel cell type). This efficiency is mainly ensured by the method of energy transformation being direct with no intermediate levels (heat and mechanic energy), unlike in case of steam power plants, combustion engines or turbines, for example. [9]
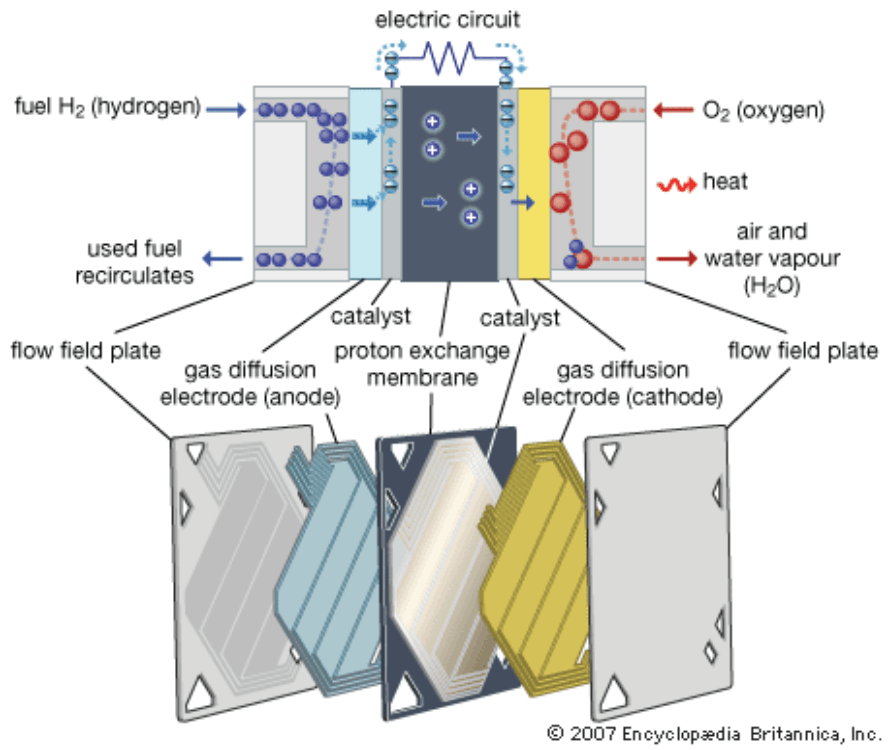
There are currently six types of fuel cells under development and these differ by electrolyte composition, operation temperatures and the type of fuel used. Proton Exchange Membrane (PEM) fuel cells are cells with polymeric electrolyte membrane known for their high conductivity, which allows for their design of light weight and reasonable dimensions. These cells use the electrolyte able to conduct H+ ions from anode towards cathode. The electrolyte used will usually be perfluorinated polymer of the sulphonic acid inserted between two electrodes impregnated with catalyser. PEM usually work under temperatures between 50 and 100 °C and the pressure of 1 to 2 bar. Figure 6 shows PEM fuel cell principle. These fuel cells provide for the chemical reaction listed below:
- Anode: 2 H2 ⇒ 4 H+ + 4 e–
- Cathode: O2 + 4 e– + 4 H+ ⇒ 2 H2O
The H+ ions pass through the electrolyte, from the anode towards cathode, whereas electrons are forced to pass from the anode towards cathode through the external electric circuit. Water produces by the cell, aggregated on the cathode, must be drained out of the cell continuously. [9]
The output of both described devices is directly dependant on the surface of electrode with main impact on their total cost. The total amount of the accumulated energy depends on the size of hydrogen container and the output from RES, i.e. the photovoltaic system.
The stage of testing of specific parts of the storage system built will be followed by their linkage to form a working system. The main components of this system comprise the renewable energy source based on the photovoltaic technology, the hydrogen production block with electrolyzer and the container for storage of hydrogen produced and the production electro-block with fuel cell modules and semi-conductor invertors, current inverters respectively.
Measurements on Fuel Cell System
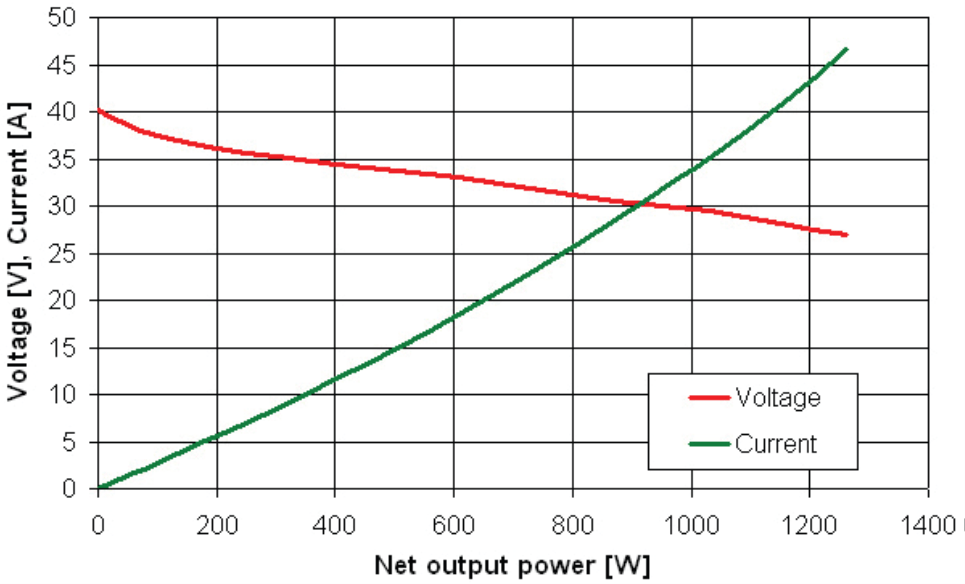
Our research laboratory is equipped with two low-temperature fuel cells Nexa Power Module (Ballard Power Systems Inc.). Rated power output of one Nexa Module is 1200 W. They are being tested in various operating conditions. The measuring procedure consists of cyclic measurements of the load characteristics of NEXA, whereas the system is connected to the distribution network via inverter, in between these measurements, to supply the electric energy. The load characteristics you can see on Figure 7. That is demonstrated in the fuel consumption graph (see Figure 8). This fuel consumption has been determined from the flow meter after the so called purge of cells. This purge deprives cells of impurities and water on regular basis, as those are accumulated on electrode surfaces to intercept the electrochemical reaction.
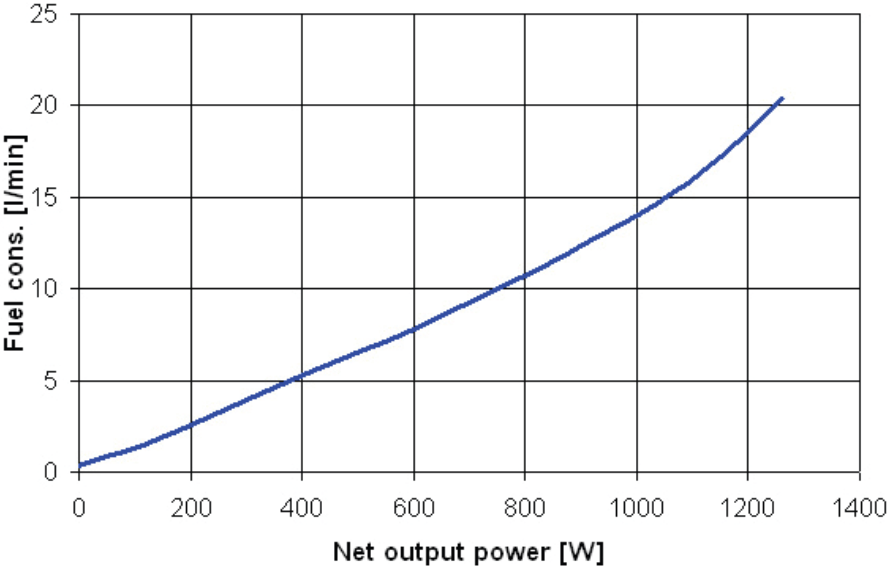
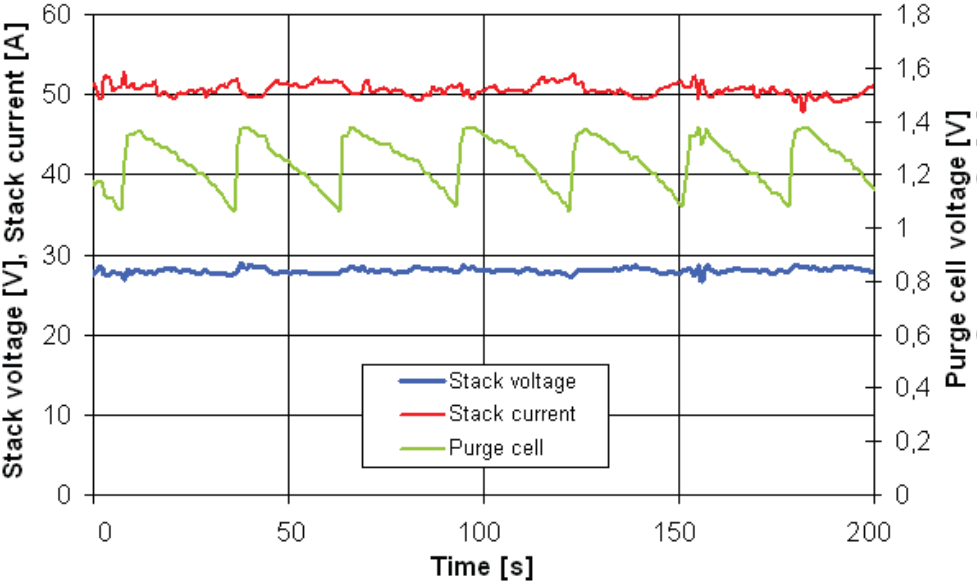
The frequency of purges rises with the increase of cell power output. The NEXA power system measures the voltage over two cells within a stack (fuel cells connected in series), the so called purge cells, to conduct the purging of fuel cells with hydrogen once the voltage has dropped below a certain level to restore the higher voltage in cells again (see Figure 9).
The fuel used for cleaning is drained out of the system unused for the reaction within fuel cells. Yet it shall be included into the overall consumption. That was the reason why we conducted accurate measurements within the system under electronic load at the particular nominal power output, one hour for each. The consumption was determined using a flow meter with an integration member. The Figure 9 shows the normal curve of stack values during the operation.
Wind-Hydrogen Power Plant
This is a complex combining a wind power plant and the hydrogen storage system based on the Utsira island at the Western coast of Norway. The entire complex is fully autonomous. The insular power distribution network supplies electric power to every household on the island, whose annual consumption amounts to approx. 200 MWh. Figure 11 shows the photograph of the entire complex with the evident tube of wind power plant with the installed capacity of 600kW, with the container and electrolyzer providing the installed capacity of 48kW situated below, with the hydrogen production capacity of 10m3/h, and the compressor with the output of 5,5kW. The photograph shows the container for 2.400 m3 of hydrogen compressed at the ratio of 200 bar. There are containers housing he fuel cell providing the output of 55kW on the right hand side. Both the electrolyzer and the fuel cell are of PEM type. [10]
When the wind turbine at Utsira is running at optimum level, it will produce more energy than the consumption is.

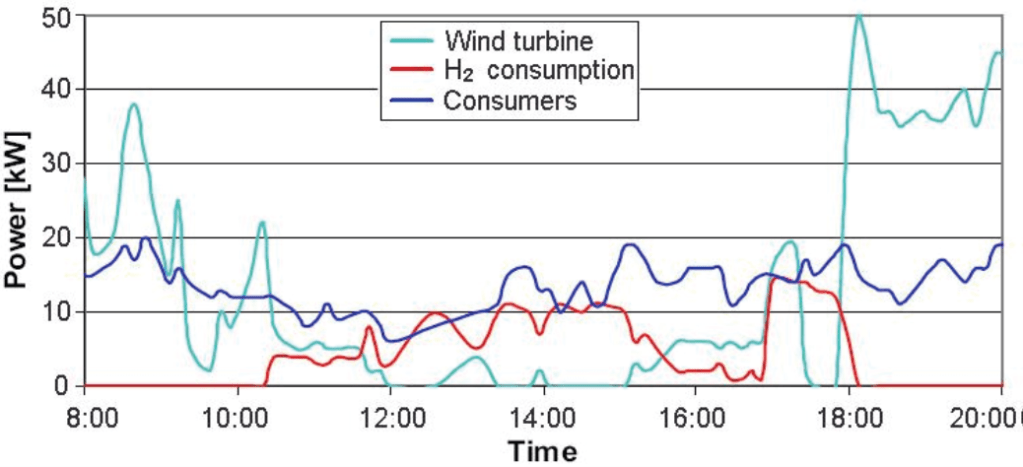
The surplus energy is used to produce hydrogen through water electrolysis. The hydrogen produced is compressed and stored in a gas storage vessel and is available when needed. Under circumstances when the wind turbine is not in operation (i.e. when there is too little or too much wind) the hydrogen is used in a fuel cell and a combustion engine-generator unit to produce power. Example of operation of power plant at Utsira is illustrated on Figure 11. On this graph you can see that the wind power decreases and cannot supply the demand. In this period the fuel cell and engine-generator are started and are balancing the load. [10]
Conclusion
The accumulation of electric power, especially the power gained from the photovoltaic and wind power plants, is of significant importance with respect to their operation in relation with the electric power system. Those are sources providing variable and unreliable supply of electric power over time, which has negative impact on the operation of the electric power system. The accumulation of electric power produced by those units can contribute towards substantial reduction of the control power, which shall be maintained within the electric power system. There is a whole range of suitable accumulation technologies available. However, their practical application in relation with the RES mentioned is still subject to research in progress.
Redox flow batteries are seen as a prospective technology as their accumulation capacity is limited by the amount of liquid electrolyte within only, i.e. the size of magazines used. These can be used to build large accumulation facilities within electric power networks. Construction of a CAES will be mainly dependant on the local geological conditions. This system is very complex from the implementation point of view, same as the pumped-storage hydro plant. There are very few CAES pilot projects in operation only.
The hydrogen system can be used to provide for accumulation of electric power in large amounts as well.
The higher cost of implementation might still represent certain problems as those are determined by the cost of materials used. However, this technology is subject to continuous dynamic development, whose future lies not only in stationary applications yet even in mobile projects. The accumulation system mentioned above can be supplemented with flywheels or super capacitors able to cater for short-term fluctuations in supply of electric power.
This work is supported by The Ministry of Education, Youth and Sports of the Czech Republic, project CEZ MSM6198910007″.
REFERENCES
[1] Cenek, M., Accumulators: from principle to practise. FCC Public, Praha, Czech Republic, 2003
[2] NAS batteries [online],
[3] Hradílek, Z., Moldřík, P., Šebesta, R., Storage of Electric Energy Gained from Renewable Sources. Proceedings of AsiaPES, 2009, Beijing, China
[4] About Flow battery [online],
[5] Moldřík, P., Hradílek, Z., Chválek, R., Research of Energy Storage Gained from Renewable Sources. Proceedings of Elnet, 2009, Ostrava, Czech Republic
[6] CAES system [online],
[7] De Boer, P., Raadschelders, J., Flow batteries. Leonardo Energy website [online],
[8] Hradílek, Z., Chválek, R., Research accumulation of energy from renewable energy sources in fuel cells. Proceedings of EPE, 2009, Kouty nad Desnou, Czech Republic
[9] Larminie, J., Fuel Cell Systems Explained. John Wiley and Sons Inc., 2003, Chichester, United Kingdom
[10] Nakken, T., The Utsira wind-hydrogen system: operational experience. Proceedings of EWEC, 2006, Athens, Greece
Authors: prof. Ing. Zdeněk Hradílek, DrSc., Ing. Petr Moldřík, Ph.D., Ing. Roman Chválek, Technical University of Ostrava, Faculty of Electrical Engineering and Computer Science, Department of Electrical Power Engineering, ul. 17. listopadu 15, 708 33 Ostrava – Poruba, Czech Republic, E-mail: zdenek.hradilek@vsb.cz; petr.moldrik@vsb.cz; roman.chvalek@vsb.cz
Source & Publisher Item Identifier: PRZEGLĄD ELEKTROTECHNICZNY (Electrical Review), ISSN 0033-2097, R. 87 NR 2/2011